| Author |
Topic: URGENT Bisulfite/MD-P2P
Question... |
sublime
Hive
Bee |
 posted 01-10-2000 05:32 PM
posted 01-10-2000 05:32 PM
I dreamt this all last night...it was weird as fuck, but lemme describe
it:
the benzo-wacker was performed flawlessly, and then all solvent was
distilled off, leaving ~200 mL of dark goop which left a VERY neon green
residue on flask walls...greener than ever before. It was seeming like my
best dream yet. (I prestirred catalyst!) Anyhow, that mix was put in the
freezer until a couple days later. It was added to 1000 mL of saturated
dH20/bisulfite. I left it to stir and went to bed. By morning, there were
TONS of crystals! they definitely looked like chicken fat on close
inspection, but looked like green goop when viewing the whole mass of
them. I filtered the crystals out of the water. Now I have
green/chickenfat goop crystals.
How do I clean them up to look only like chicken fat? I assume that
there's some hydroquinone in there, or something. Which solvents would be
ideal for washing them? I have DCM, IPA, dH20, and Acetone.
Once washed, the crystals will be placed into a sep funnel with 10%
HCl...this should liberate the ketone, right? I've read conflicting
reports about this issue...some say 10% NaOH. Which works here?
----end of dream here---
It's weird when I ask myself questions in
my dreams...dontcha think?
Thanks all!
Ciao...
sublime
|
Sploofer
Hive
Bee |
 posted 01-10-2000 05:49 PM
posted 01-10-2000 05:49 PM
try using your head the fur will do amazing things to selectivly eliminate
oils
|
Osmium
PimpBee |
 posted 01-11-2000 04:52 AM
posted 01-11-2000 04:52 AM
I hope you filtered away the black crap as good as possible? I'm not sure
but it's possible the bilsulfite might be incompatible with
hydroquinone/benzoquinone.
Try the washing on a small scale first. I always preferred NaOH to
decompose the bisulfite, because of smell reasons, but that is up to you.
NaOH might be better because you have to wash it with NaOH anyway to
remove the hydroquinone. Divide your chicken fat into several small
portions and try some different workups. Don't forget to post your results

|
APOPLEX
Hive
Bee |
 posted 02-01-2000 12:57 PM
posted 02-01-2000 12:57 PM
I had a question, Osmium:
I worked up a SRV/KRV Wacker ala TS II. I got a red-brown-yellow liquid
after removing the
DCM.
Then I made a soln. of Na2S2O5 in dH2O. I know that this WILL give a
aq. soln. of NaSO3 in balance like this:
S2O5- + H2O <----> 2 HSO3-
So, the sat. aq. soln. should be Ok to meet the ketone I supposed in my
above liquid.
I dripped some of my liquid into the stirring bisulfite soln. I stirred
it about one hour, but NO crystals formed. Only a yellow turbid liquid
wich seperates hardly.
The SRV-Air-Wacker went exactly like described, so I`m relatively sure
that the above oil should contain a lot of ketone.
Wrong bisulfite soln. ? What do you think ?
Please help !
-------------------
APOPLEX
|
Osmium
PimpBee |
 posted 02-01-2000 01:58 PM
posted 02-01-2000 01:58 PM
Maybe too diluted? Too much bisulfite solution? I don't know, it should
work.
|
Bankrobber
Hive
Bee |
 posted 02-02-2000 01:17 AM
posted 02-02-2000 01:17 AM
hey sublime, did you wash w/ NaOH(preferably 2-3 times, especially if your
new to this ("thing") I,ve personally used bisulfate on filtered,
washed(dh2o,NaOH,sodium bicarb, and NaCl), and distilled ketone(when I was
in the "beginning", and needed that xtra assurance that I didn't fuck
anything up), if you don't do that I suspect that the contaminants can
interfere w/ the bisulfate addition(regardless of "promotions" in ts2, by
the big guy), I figure from practical exp. that this will not work on
contaminated NOT-ketone(I mean by saying this that how often do thigs ever
work out when impurities, especially the ones in NOT-ketone prod. are
there and the solution is a dirty ass' black, I mean to say it sounds
half-ass to me to assume that you aren't going to run into problems, I
don't mean that your half-ass SUBLIME, I meant a half-ass bisulfate
addition attempt). Anyways one more thought that might be of service dude,
your end result of the legal reaction should be way more clear-yellow than
a real green(end product should only have hint of green, along with
predominate clear yellow). just swish around in see-through container, and
no matter wether purified or not you'll see on glass and the solution
closest to the glass a slight yellow, that's when you know you've done
well( just practical, your original ingredient is clear and know ther's a
bunch a' yella, so ya' know your good deed for the day that you promised
mom you'd do is now done), take care dude, EVIL ASS' BANKROBBER
|
equarius
Hive
Bee |
 posted 02-02-2000 04:05 AM
posted 02-02-2000 04:05 AM
BR; So your BISULPHATE addition product reacted just fine? What yeilds?
105%? Did you use NaMetaBiSULPHATE or NaBiSULPATE or NOT some NOT other
NOT BiSULPHATE /NOT?
Apoplex; Add some EtOH to the solution and it should crash out the
crystals.. especially when comming from SRV reactions. And make sure the
solution is completly saturated.
Disclaimer: Fester (the truth fairy) made me do it.
|
sparks
Hive
Bee |
 posted 02-02-2000 04:24 AM
posted 02-02-2000 04:24 AM
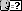
Sodium bisulfite
Synonyms: Hydrogen sulfite sodium; Sodium acid sulfite; Sodium hydrogen
sulfite; Sulfurous acid, monosodium salt; Monosodium sulfite; Sodium
sulfite (NaHSO3)
Not Na2S2O5.
|
Osmium
PimpBee |
 posted 02-02-2000 04:48 AM
posted 02-02-2000 04:48 AM
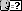
Solid NaHSO3 does not exist. When evaporating NaHSO3 solutions Na2S2O5 is
formed. This is the correct chemical.
|

 the
Hive BB
the
Hive BB 
 Methods
Discourse
Methods
Discourse 
 URGENT
Bisulfite/MD-P2P Question...
URGENT
Bisulfite/MD-P2P Question...